SARS-CoV-2 Neutralization Antibody Detection ELISA Kit (TBS3240)
$489.00
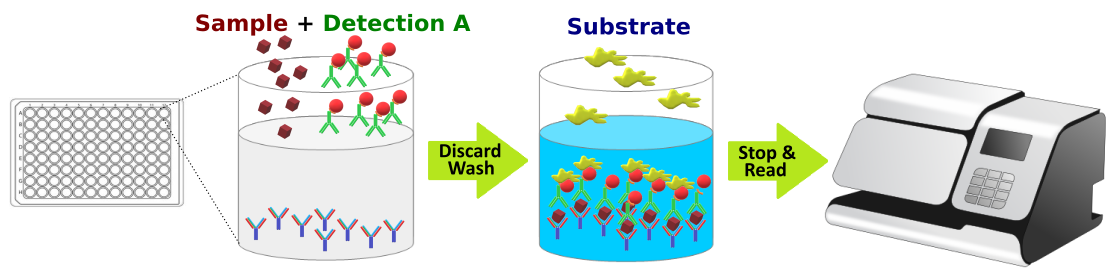
The TribioScience SARS-CoV-2 Neutralization Antibody Detection ELISA Kit employs the principle of competitive ELISA to detect the presence of anti-SARS-CoV-2 antibodies in serum/plasma samples.
| SKU | Stock | SIZE (kit) | Price | Quantity | ||
|---|---|---|---|---|---|---|
| TBS3240 | Yes | 1 x 96-Well Plate | $489.00 |
- Description
- Terms
- Additional information
- Documents
- Reviews (0)
Description
The ELISA assay is based on the competitive principle. Each well of the supplied microtiter plate has been pre-coated with recombinant SARS-CoV-2 Spike RBD-hFc protein. When HRP conjugated recombinant human ACE2 protein is added simultaneously to wells along with diluted human serum/plasma samples, immobilized RBD will bind human ACE2 protein. The binding of RBD to human ACE2 protein is dose-dependently inhibited by the presence of anti-SARS-CoV-2 human neutralization antibodies in serum samples. Following the sample incubation step, unbound ACE2-HRP conjugate is washed away. A Positive Control is a known human antibody (IgG1 subtype) that is specific for SARS-CoV-2 spike protein with a binding epitope for RBD binding site. The positive control antibody is validated by an established pseudovirus infection of cultured human cells for proven neutralization function. Human IgG1 isotype control is used as a Negative Control sample. The absorbance of the unknown sample can then be compared to the Positive and Negative controls to determine the absence or presence of anti-SARS-CoV-2 human neutralization antibodies. In the competition assay, the greater the amount of anti-SARS CoV-2 human neutralization antibodies in the sample, the lower the color development and optical density reading.
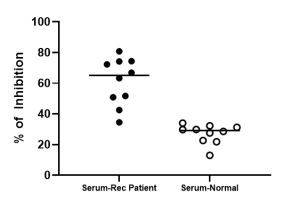
Specificity: This kit is for the detection of Human anti-SARS-CoV-2 neutralization antibodies.
Main Features
- Simple: The sample and HRP detection combined into one step
- Fast: Just 1-hour incubation and 1step washing
- High sensitivity: detection of 7.0 ng/mL
- High specificity: Specifically detecting human ant-SARS-CoV-2 neutralization antibodies.
Sample Types: This kit is recommended for use with human serum/plasma. Use with other sample types like SARS-CoV-2 immunized animal serum is possible but not validated in this testing kit.
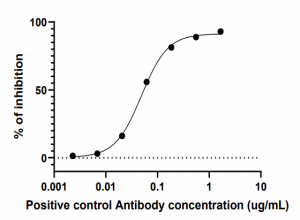
Detection Range: 10 to 500 ng/ml using positive control antibody 8A5
Sensitivity: Typically, about 7.0 ng/mL with IC50 at approximately 50.0 ng/mL
Performance: Intra-Assay CV (< 10.0%); Inter-Assay CV (< 15.0%)
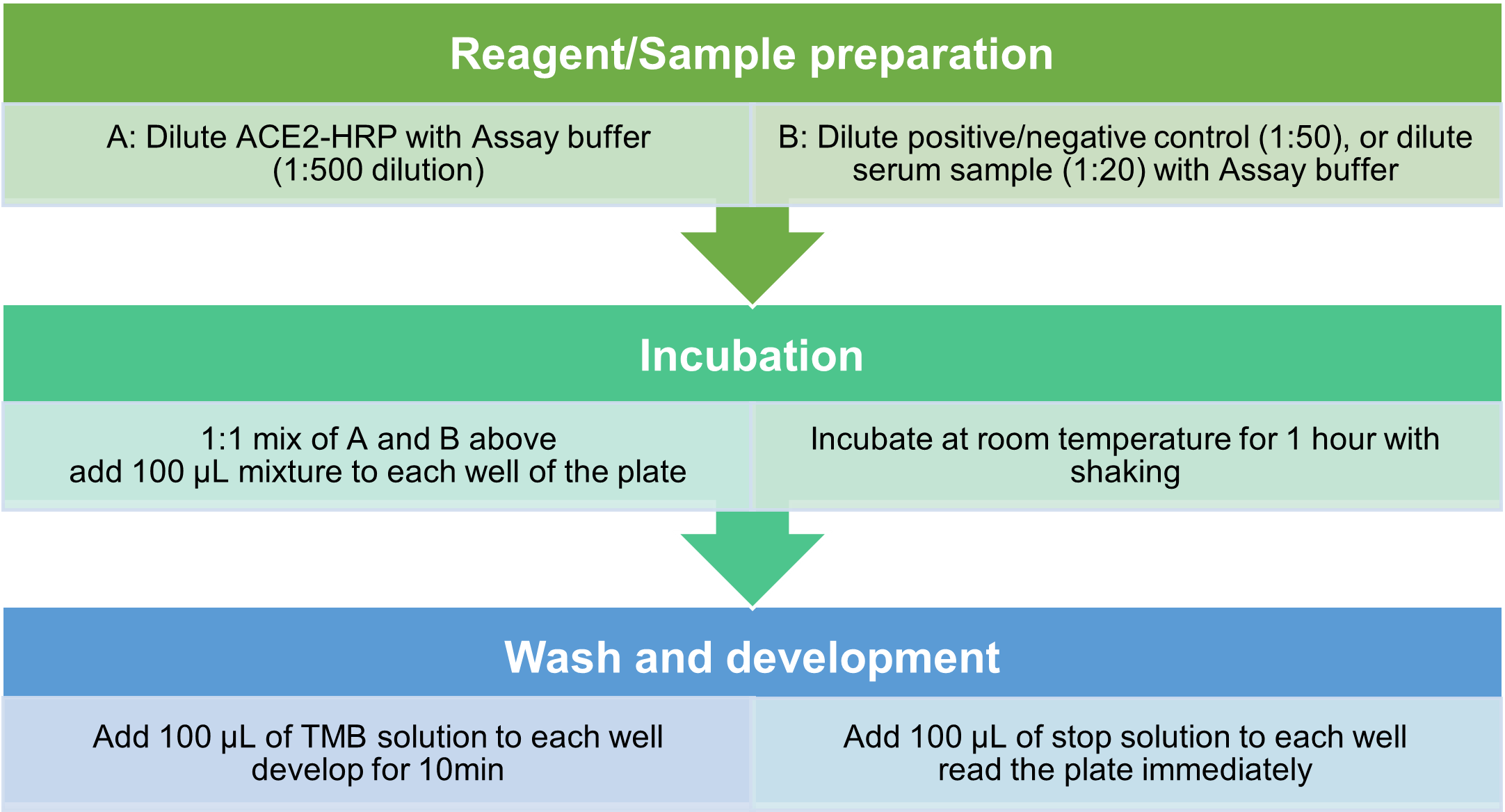
Assay Protocol
Typical Data
Typical Data
Additional information
| SIZE (kit) | 1 x 96-Well Plate |
|---|
Be the first to review “SARS-CoV-2 Neutralization Antibody Detection ELISA Kit (TBS3240)”
You must be logged in to post a review.













Reviews
There are no reviews yet.